New Faculty at UW Working on Climate Science and Sustainability

Julie Rorrer, Assistant Professor, Chemical Engineering

Jungwon Choi, Assistant Professor, Electrical and Computer Engineering

Zachary Sherman, Assistant Professor, Chemical Engineering

Claire Willing, Assistant Professor, Environmental and Forest Sciences

Kendall Valentine, Assistant Professor, Oceanography

Corey Garza, Professor, Aquatic and Fishery Sciences

Michelle Muth, Assistant Professor, Earth and Space Sciences

Sameer H. Shah, Assistant Professor, Environmental and Forest Sciences

Vikram Iyer, Assistant Professor, Computer Science & Engineering

Cory Struthers, Assistant Professor, Public Policy and Governance

Jeremy J. Hess, Professor, Atmospheric Sciences

Shijing Sun, Assistant Professor, Mechanical Engineering

June Lukuyu, Assistant Professor, Electrical and Computer Engineering

Christie Hegermiller, Assistant Professor, Civil and Environmental Engineering

Celina Balderas Guzmán, Assistant Professor, Landscape Architecture

Bhuvana Srinivasan, Associate Professor, Aeronautics and Astronautics
View More New Featured Faculty
![[Concept - solving a complex problem. Blue maze and floor with yellow solution path with arrow. Banner size.] [Concept - solving a complex problem. Blue maze and floor with yellow solution path with arrow. Banner size.]](https://www.washington.edu/research/wp-content/uploads/AdobeStock_502554360-1-copy-scaled.jpg)




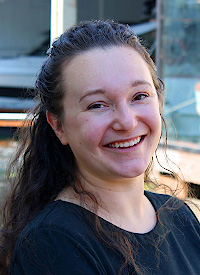










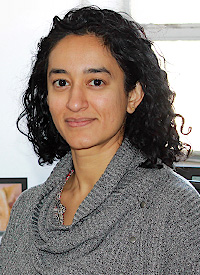
![[Student researchers at the Molecular and Cellular Biology Lab] [Molecular and Cellular Biology Lab]](https://www.washington.edu/research/wp-content/uploads/2017/04/uw-research-feature-uwtoday.jpg)
![[UW Faculty, 1882] [1882-faculty]](https://www.washington.edu/research/wp-content/uploads/1882-faculty.gif)