Human Subjects Division
Research Involving Seattle Children’s
Research involving two institutions normally requires review by each institution’s IRB. To avoid this “dual review” and the extra work it requires from researchers, Seattle Children’s (SC) and the UW IRB have a reliance agreement that describes the circumstances in which one of the IRBs will conduct the review on behalf of both institutions.
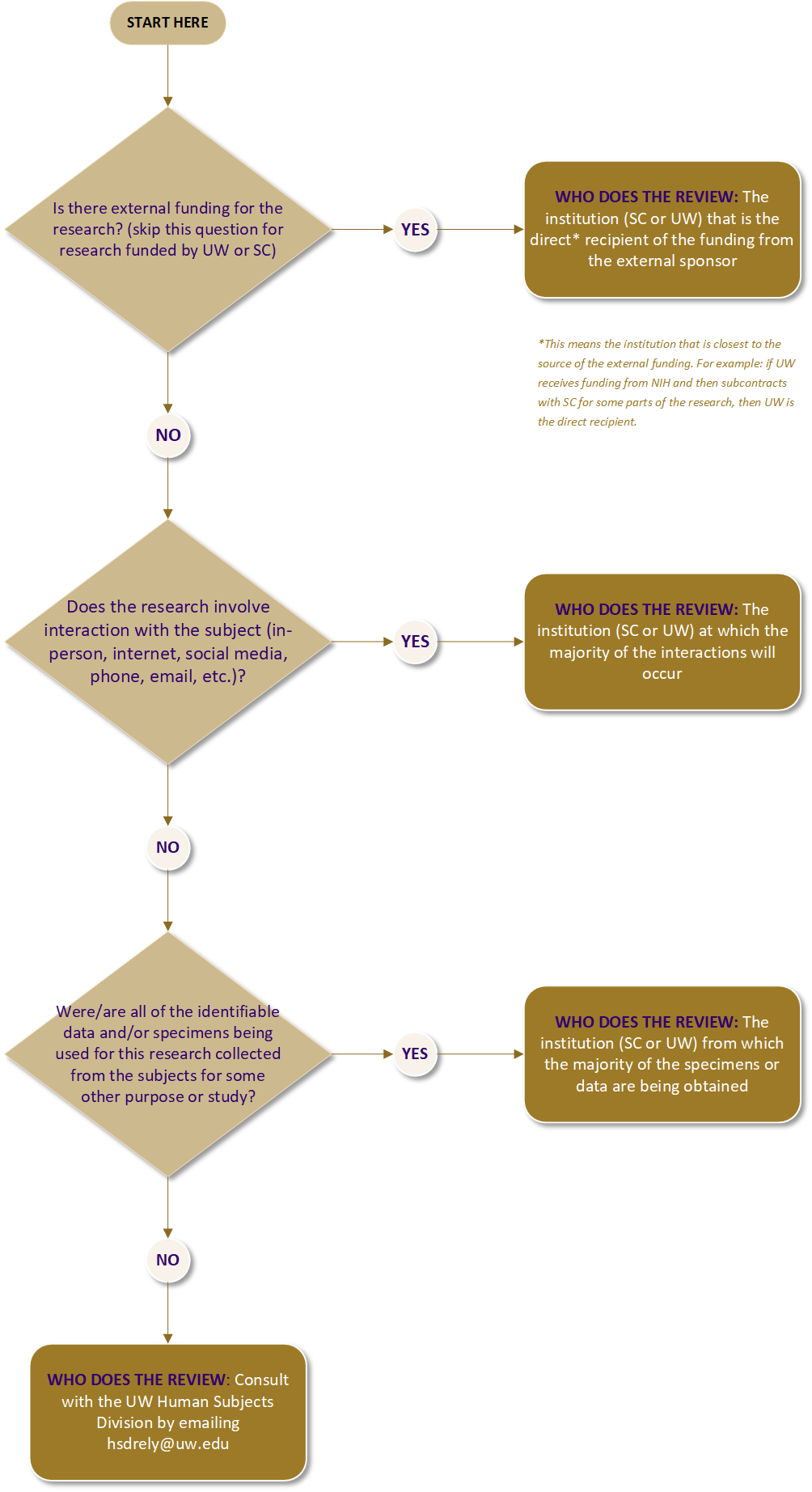
Use the flow chart below to determine which IRB should do the review. If you have any questions, consult HSD at hsdrely@uw.edu.
Before you begin
- Assess whether both institutions are engaged in the research. If only one institution is engaged, you should submit to that institution’s IRB.
- Use the engagement guidance to identify whether UW is engaged in the research.
- Consult with the Seattle Children’s IRB to identify whether SC is engaged in the research.
- Assess whether another IRB is the right option. For example, most industry-sponsored research at UW should be reviewed by an independent IRB (for example, WCG IRB) and some cancer-related research should be reviewed by the Fred Hutch IRB or the NCI IRB. Review the information about identifying the correct IRB if you are unsure.
What to do if SC will do the review
- Submit an External IRB Application in UW’s Zipline system to request HSD’s formal authorization for SC to review the research. HSD will evaluate your request and provide you with a written authorization letter.
- The SC investigator should submit an application to the SC IRB using SC’s forms and process. Be sure to attach the HSD authorization letter; the SC IRB will not review the application on behalf of UW without this letter.
- After the SC IRB has approved the UW’s participation, send the approval letter to hsdrely@uw.edu.
- Obtain all other applicable ancillary reviews (consult the Checklist of Researcher Responsibilities for Externally Reviewed Studies) from the usual UW offices and SC as needed. Examples: Financial Conflict of Interest (FCOI) review; Radiation Safety review.
- Exception: SC will provide Genomic Data Sharing (GDS) certification (if necessary) on behalf of both institutions.
What to do if UW will do the review
- Submit a UW IRB Application in the Zipline system. Be sure to:
- Describe all involvement of SC in your application (the Zipline SmartForms as well as the IRB Protocol form that is uploaded).
- Follow the instructions for the UW Reviewing for Other Institutions pathway.
- On the Study-Related Documents SmartForm: Upload a completed SUPPLEMENT: Multi-site or Collaborative Research to Question 3.
- Request SC’s authorization to rely on the UW IRB by submitting an External IRB Application in the SC Click IRB system. SC will evaluate your request and provide you with written authorization for the UW IRB to do the review. Deliver this letter to hsdrely@uw.edu.
- The UW IRB will review and approve the overall protocol. This is not yet approval for the involvement of SC. After the UW IRB has approved the overall protocol, the HSD Reliance Team will work with you to approve the addition of SC to the overall application.
- After UW IRB approval for SC has been obtained, you are responsible for:
- Informing the SC IRB of UW’s approval for SC.
- Informing SC investigators about any conditions of the IRB approval.
- Informing SC investigators about their obligation to comply with UW IRB reporting requirements about noncompliance, complaints, problems, etc., although such reports must be submitted by you to the UW IRB.
- SC investigators remain responsible for obtaining any applicable ancillary review from SC (for example, Financial Conflict of Interest (FCOI) review, Radiation Safety Review).
- Exception: UW will generally provide Genomic Data Sharing (GDS) certification (if necessary) on behalf of both institutions.
SC or UW IRB Decision Tree