Human Subjects Division
Modify Approved Multi-institutional Study
Table of Contents
Before You Begin
A study-level modification is used when a change applies to the overall study, including all participating sites. This also includes changes that affect both the study and its sites.
A site-level modification is used when a change applies to one specific participating site only and does not affect the overall study or other sites.
In multi-site studies, modifications may be required at the study level, the site level, or both, depending on the change requested and how the study is set up in Zipline.
Start by determining the scope of the change:
If the change applies to the overall study, submit a study-level modification. For more information:
If the change applies to one specific site only, submit a site-level modification. For more information:
If the change affects both the study and one or more sites, submit a study-level modification first.
HSD strongly recommends waiting to submit any sit- level modifications until the study-level modification has been reviewed. During review, HSD staff will evaluate whether a site modification is also required and will provide instructions on when and what to submit.
Because requirements can vary based on how the study is configured in Zipline, it may not always be clear which modification is needed. If you are unsure, contact hsdrely@uw.edu for guidance before submitting.
Steps for Study Modifications
The steps for multi-site study-level modifications are the same as the steps for single-site study modifications. Go to Modifications to UW Reviewed Studies for detailed steps.
Steps for Site Modifications
Site modifications apply to a single participating site. If HSD requires changes, they will withdraw the modification because clarification requests are not available for site modifications. The PI or PI proxy must resubmit the modification before review can continue.
- In the SITE workspace, click Create Site Modification
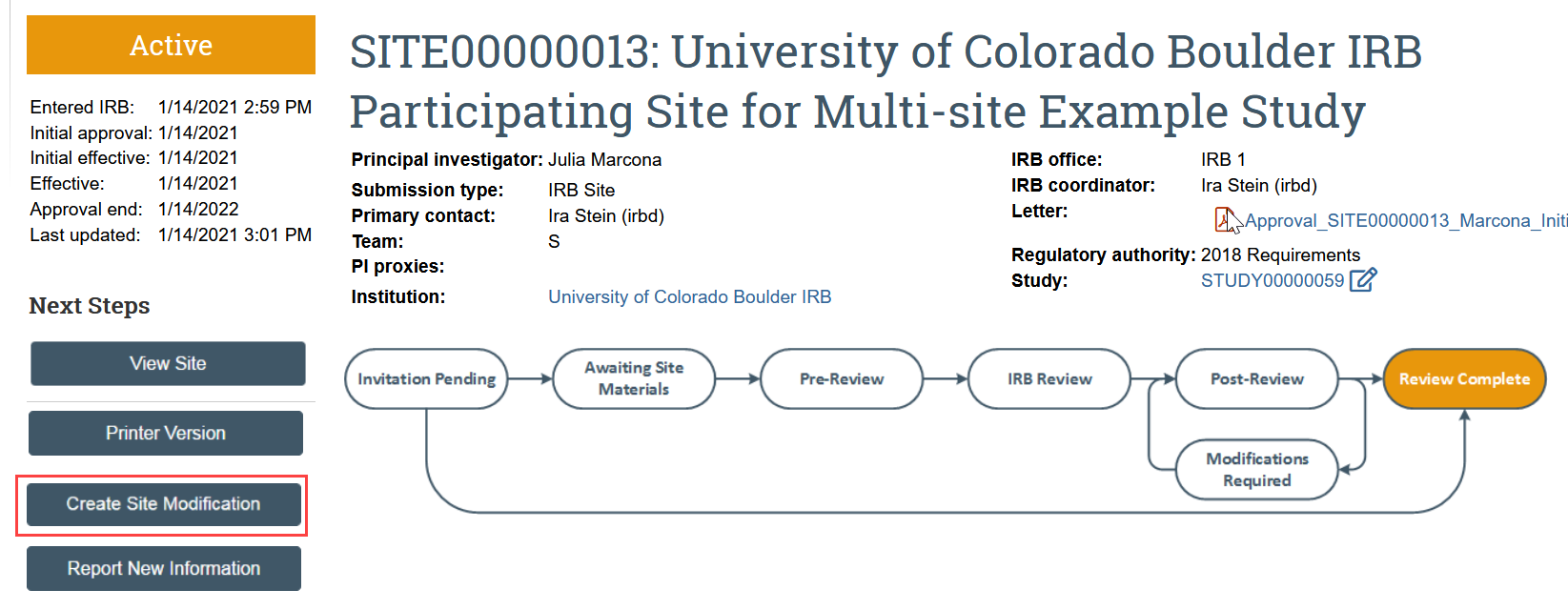
- You are now in a draft version of the site submission and should be on the Basic Site Information page. Update the draft submission to reflect the proposed modification by revising the Site SmartForms as needed and uploading any new or updated documents.
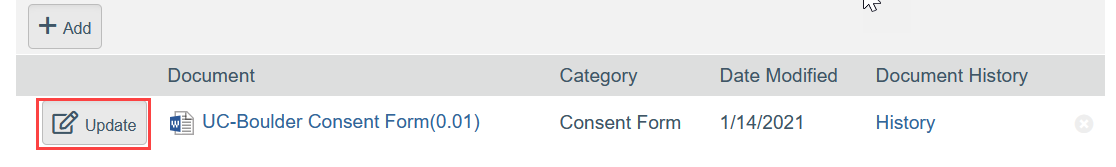
- To Update a Document:
- Click Update
- Select the revised file from your computer
- Click OK in the Edit Attachment window
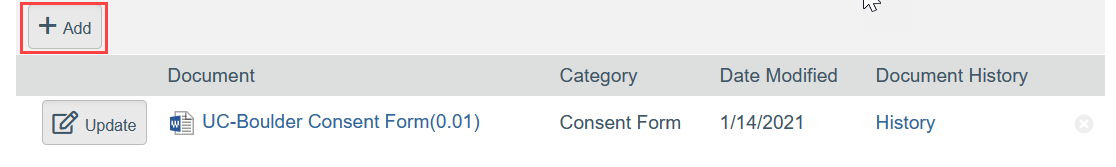
- To Add a New Document:
- Click Add
- Select the new document from your computer
- Click OK in the Edit Attachment window
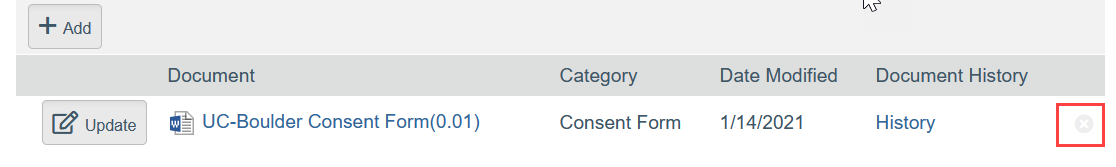
- To Remove a Document:
- Click the X by the document
- Click Save and Exit when you are finished editing the modification
- Click Submit in the modification workspace. The Submit activity must be completed once the modification is ready to go to HSD.
- Click OK to provide required verifications
The modification transitions to Pre-Review state and is now in HSD’s queue for review.
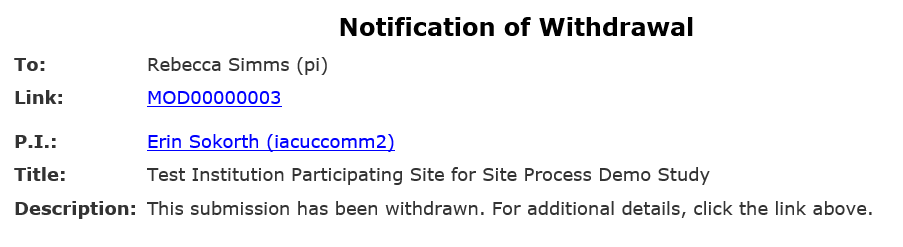
Important: Site modifications do not have a formal clarification request process. If HSD needs additional information or changes before approval, they will withdraw the site modification and return it to Pre-Submission with reviewer comments. You must address the requested changes and resubmit the modification for HSD to continue review.
- When the submission is withdrawn, an automatic notification is sent to the PI, any PI proxies, and the parent study’s primary contact.
- Select Edit Modification and make any needed changes to the application
- Select Submit to send the site modification back to HSD for review
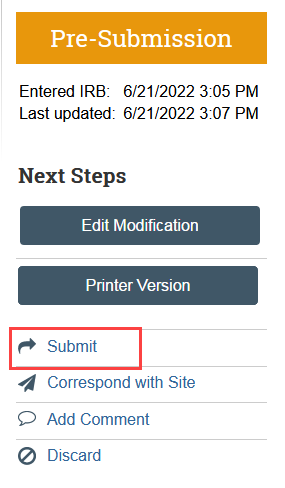
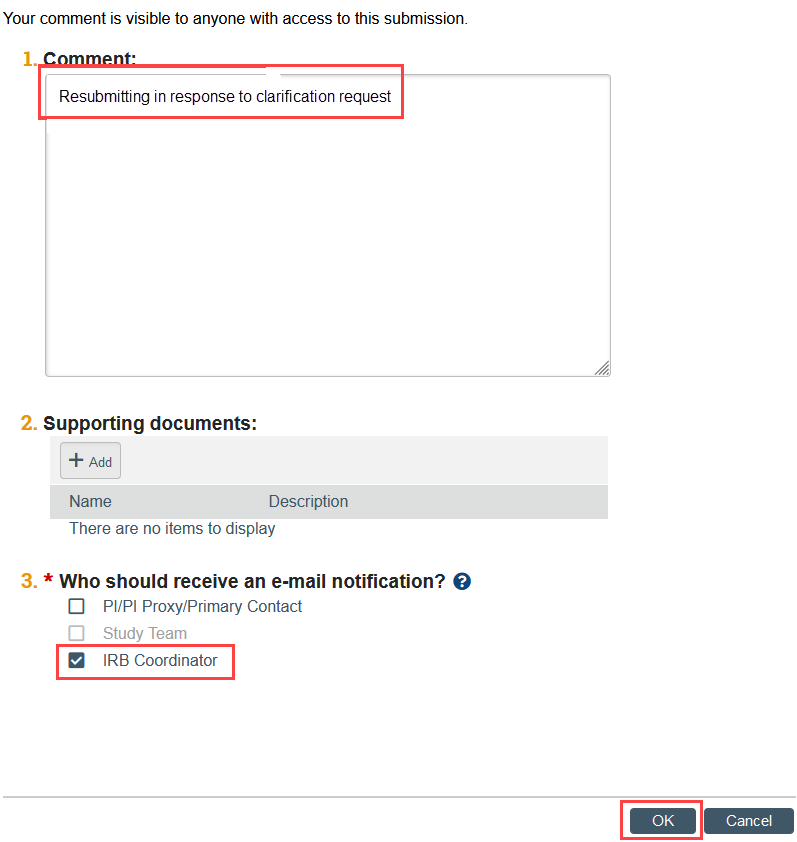
- Click Add Comment in the modification workspace. Indicate that you have provided the response to clarification, check the box to notify the IRB coordinator, and click OK. This ensures that the correct IRB coordinator is notified.
After Modification Approval
After the modification is approved, the changes are published in the site record, so the site always contains the currently approved information. The UW PI, PI proxy, and primary contact also receive a notification containing the modification approval letter.

