May 22, 2000
UW partners with Advanced Tissue Sciences and others in $10 million grant to ‘grow’ human heart tissue
Bioengineering researchers at the University of Washington will lead a multi-million-dollar effort to grow functional human heart tissue, an undertaking that could lead to tissue-engineered replacement hearts and set in place the technology to grow other major organs in the laboratory.
The project, funded by a grant from the National Institutes of Health for $10 million over five years, will initially focus on culturing thick “patches” of cardiac muscle that could be grafted onto damaged hearts to improve their efficiency.
The next step will be engineering a “ventricular tube,” a cylinder of rolled cardiac muscle with valves that could assist a weakened heart with pumping. Then a full ventricle will be attempted. Eventually, researchers hope their work will enable scientists to grow a fully functional human heart.
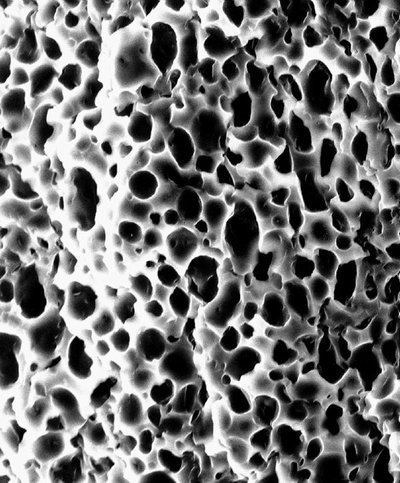
In growing such devices, and eventually an entire heart, researchers will begin with a “scaffolding,” or porous structure upon which cardiac cells can gain a foothold. The scaffolding is seeded with cells then placed in a bioreactor, which maintains a steady temperature conducive to development and provides cells with the nutrients they need to grow and reproduce. Advanced Tissue Sciences‘ (NASDAQ:ATIS)patented technology will play a key role in the partnership’s efforts.
Initially, researchers will be working with skeletal muscle cells, or myocytes, as well as with cardiac cells. Stem cells, or cells that have the ability to develop into specialized cells such as cardiac cells, provide another important key. The use of stem cells on a scaffold is largely unexplored and will be a major thrust of the project. Such cells could give scientists a virtually never-ending supply of cardiac cells for tissue engineering. And they may even hold clues to solving the problem of rejection by using the patient’s own cells to grow new organs.
The effort represents a public-private collaboration led by the University of Washington. Partners, who will split the $10 million award, include Advanced Tissue Sciences of La Jolla, Calif., the Hope Heart Institute in Seattle, Advanced Polymer Systems of Redwood City, Calif., and the University of Toronto. The team comprises 44 scientists working in nine UW laboratories and five full labs at the partner companies. The project is set on a 10-year timetable, and the group will apply to renew funding at the end of the five-year grant.
“This is a sort of mini-Apollo mission,” said principal investigator Buddy Ratner, bioengineering professor and director of the University of Washington Engineered Biomaterials program. “Their goal was to walk on the moon in 10 years. We also have an exciting, almost audacious goal – to grow a human heart ventricle.
“We have a team of people who are tops in their field who will be working simultaneously on different aspects of the mission. The heart is a complex organ, which makes for a complex project, so we have a complex team. We’ll be coordinating our researchers’ results as we go, moving ahead as we break new ground.”
Breaking new ground will be an integral – and exciting – part of the effort, according to Kip Hauch, faculty member in the UW Department of Chemical Engineering and project coordinator.
“This field is moving so quickly, it’s changing every week,” Hauch said. “We’ll have to stay on the leading edge, taking advantage of advances in both biology and engineering to address the problems that need to be solved to make this work.”
The stakes are high. In the United States alone, only 2,300 hearts are available for transplant each year. That means 50,000 people who meet the strict criteria for potential heart transplants die annually for want of available organs. Many of those would be helped with a ventricular patch of cardiac tissue, a sort of “contracting living Band-Aid” that could improve cardiac performance to the point that a transplant wouldn’t be necessary, Ratner said. A ventricular tube could be similarly beneficial.
Designing the scaffolding is an early critical phase of the project. Researchers will investigate the use of various polymers and techniques to create the porous foundation, which Hauch describes as “looking like Swiss cheese” when viewed microscopically. Preliminary findings indicate that uniformity and pore size will be important in fostering tissue development.
Advanced Tissue Sciences will be a major resource in this area, Ratner said. The company holds patents for seeding cells, including stem cells and genetically engineered cells, onto scaffolds to grow three-dimensional tissue. It uses those techniques to bioengineer commercially available replacement skin products, as well as other tissues. The company also has patented technology for bioreactor systems that replicate conditions inside the body to produce engineered tissue on a large scale.
“Producing something on a small scale at the university and scaling that up to work on an industrial scale are very different things,” Ratner said. “We’ll be relying on Advanced Tissue Sciences’ expertise to develop methods to do that.”
Gail K. Naughton, Ph.D., president and chief operating officer at Advanced Tissue Sciences, said her company is looking forward to contributing its technology, experience and intellectual property to the effort.
“We have gained valuable experience in upscaling our first commercial products and have applied this knowledge to working with a variety of other cell types and bioreactor systems,” she said. “Working with leaders in the field like Buddy Ratner’s group at the University of Washington and the prestigious group of researchers participating in this project, we will together continue to advance the field of tissue engineering. With our combined efforts, the goal of tissue-engineering a human heart ventricle has the potential to become a reality.”
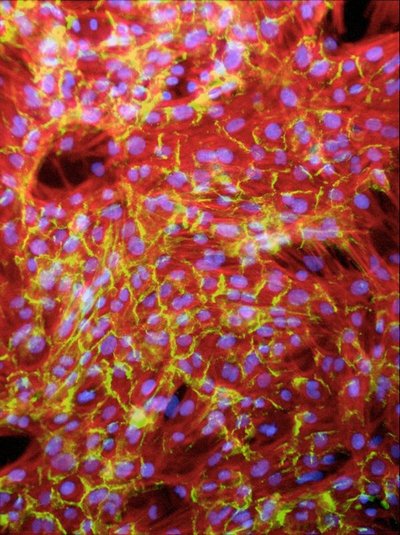
An interdisciplinary UW group led by Patrick Stayton, associate professor of bioengineering and an investigator on the project, has been able to make cardiac cells form organized fibers by arranging natural adhesive proteins on biodegradable scaffolds. Working with Chuck Murray, a UW cardiovascular pathologist, and Steve Hauschka, a biochemist, the group found that cells fall in line with the proteins, forming aligned fibers that look much like those found in natural cardiac tissue. The line of cells beat together and show evidence of intercalated disks, the connections between cells that provide mechanical and electrical junctions and allow them to communicate and coordinate with one another.
“This is the kind of preliminary research that we will be building on,” Hauch said. “We already have promising work in doing this in two dimensions, and we’ll apply that to a three-dimensional environment.”
Ratner said he knows the team faces numerous obstacles in growing living cardiac devices. The NIH recognized that as well when it awarded the grant.
“They said they realized this is a high-risk undertaking, but the enormous potential benefits more than justify the risk and they thought our team was the most likely to succeed,” Ratner said. “Personally, looking at the people involved, I like our chances.
###
For more information, contact Ratner at (206) 685-1005, (206) 616-9718 or ratner@uweb.engr.washington.edu, or Hauch at (206) 543-0289 or hauch@u.washington.edu. At Advanced Tissue Sciences, contact Naughton at (858) 713-7731.
For B-roll of beating, cultured cardiac cells, contact Rob Harrill at (206) 543-2580 or rharrill@u.washington.edu. For B-roll of tissue engineering processes at Advanced Tissue Sciences, contact Jana Stoudemire at (858) 713-7802 or jana.stoudemire@advancedtissue.com.
Advanced Tissue Sciences Disclaimer
The discussions contained in this press release relating to research, development, or commercialization of the Company’s products that are not strictly historical may be “forward-looking” statements which involve risks and uncertainties. Funding provided in subsequent years over the term of the five-year grant is contingent on the availability of funds from Congress, subject to satisfactory performance by the Company and other collaborators, and will be at the sole discretion of NIH. No assurances can be given that the Company will successfully be able to obtain continued funding when needed, develop any such products, complete clinical trials, obtain regulatory approvals (or that any such approvals will be on a timely basis), be able to manufacture or successfully commercialize any such products. These and other risks are detailed in the Company’s publicly available filings with the Securities and Exchange Commission such as Advanced Tissue Sciences’ Quarterly Report on Form 10-Q for the quarter ended March 31, 2000. The Company undertakes no obligation to release publicly the results of any revision to these forward-looking statements to reflect events or circumstances arising after the date hereof.