Because only the person who submits the RNI can respond to any requests for clarification and make edits to the RNI submission, the PAVE report should be submitted by a study team member who is able to respond to any further questions about the report, its findings, and any action plan. The PI, any PI proxies, the primary contact, and study team members of the study are given read-only access to the RNI submission. The PI, any PI proxies, and primary contact receive the same e-mail notifications as the RNI submitter throughout the workflow.
If the PAVE report will result in changes to the study, a study modification [1] should also be submitted and associated with the RNI.
Submit PAVE Report
1. Navigate to the study workspace either directly using the link provided by HSD staff or by navigating in Zipline [2]
2. From the study workspace, click Report New Information to go to the RNI SmartForm
3. Enter a short title using the following format: [PI Last Name]_PAVE Report
4. Enter the date you are submitting the PAVE report as the “date you became aware of the information”
5. Select Report as the category of new information
6. Enter a brief statement such as “See attached PAVE report” as the description of the new information
7. Indicate your assessment (question 5). Answers should be based on the observations made in the PAVE report and any proposed corrective action you submitted.
8. Confirm that the study is associated with the RNI and if the study requires revision based on the PAVE report, associate the relevant study modification [1] with the RNI
-
- If you created the RNI from the study workspace you should see the study automatically associated with the RNI
- To associate a study or modification with the RNI:
- Click Add
- Filter by the ID number for studies or Parent ID for modifications
- NOTE: Modifications can only be associated with the RNI if the parent study is associated with the RNI. If the parent study is associated with the RNI and you do not see the modification, click SAVE in the RNI SmartForm and try again.
- Select the relevant study or modification and click OK
9. Upload the PAVE report provided by HSD staff as a supporting document
10. Save and Exit the RNI SmartForm
11. From the RNI workspace, click Submit RNI and click OK to complete required verifications
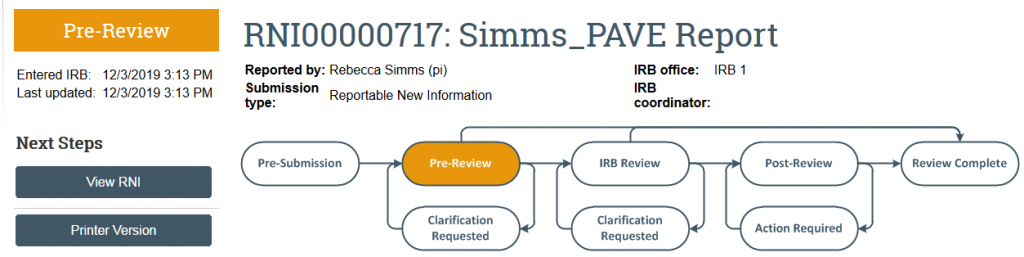
After the Submit RNI activity is completed, the RNI should then transition from Pre-Submission state to Pre-Review state. Once the RNI is in Pre-Review, it is in the HSD compliance team queue for review.