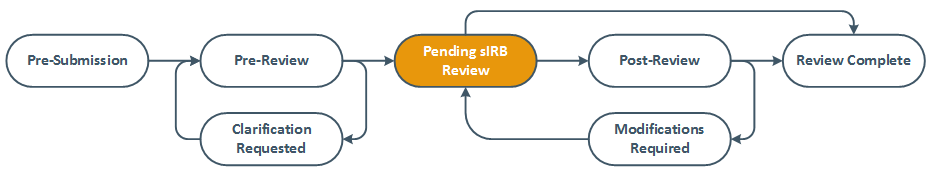
After Initial Approval
Table of Contents
UW Reviewed Studies
After HSD has made a final determination about the study, the Principal Investigator, any PI proxies, and the primary contact all receive an email notification communicating the outcome.
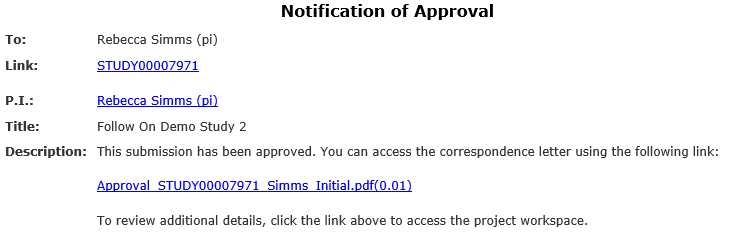
A formal letter documenting the determination and providing additional information is attached to the notification and is also available in the study workspace in Zipline.

Depending on your study, here are some additional actions you may need to take later:
- Manage access to your study
- Modify your study
- Renew or close your study
- Report new information to HSD
External IRB Studies
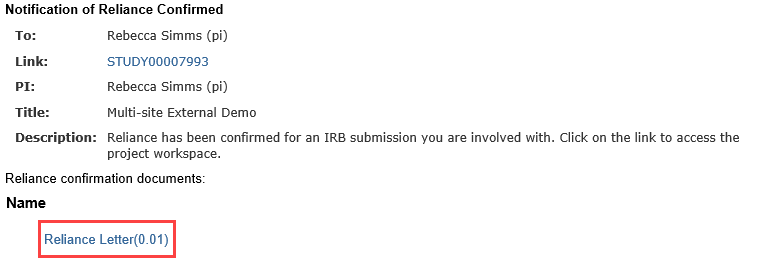
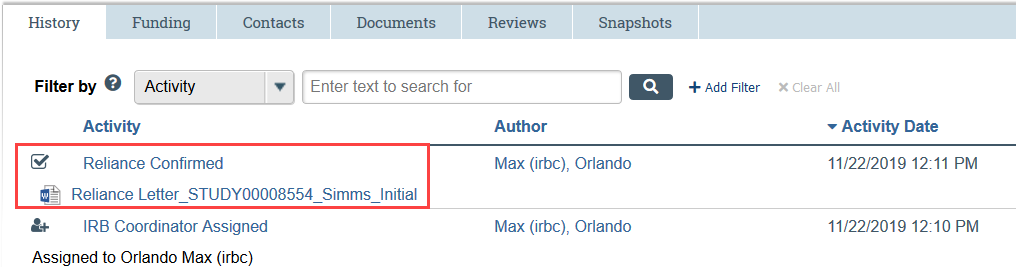
Once reliance has been confirmed, the Principal Investigator, any PI proxies, and the primary contact all receive a notification. The Reliance Confirmation Letter is attached to the notification and available in the study history.
The study will remain in Pending sIRB Review state until HSD records the reviewing IRB’s approval.
- Studies reviewed by WCG Group IRB and Advarra: Researchers typically do not have to do anything. WCG Group IRB and Advarra automatically send approval letters directly to HSD for most studies. HSD will upload these to Zipline as they are received.
- Studies review by Trial Innovation Network IRBs: Researchers do not have to do anything. These IRBs automatically provide approval letters to HSD. HSD will upload these to Zipline as they are received.
- Studies reviewed by any other IRB, including Fred Hutch and Seattle Children’s: These IRBs may or may not provide approval letters directly to HSD. If HSD receives the documents directly, HSD staff will upload the document to Zipline. However, the research team should email a copy of the approval letter to hsdrely@uw.edu to ensure that HSD receives the documents.