HSD only requires limited ongoing information about externally reviewed studies. See the CHECKLIST: External IRB, UW Researchers [2] for more information about when studies that are being reviewed by a non-UW IRB require a study update. Contact hsdrely@uw.edu [3] with any questions.
See the table below for the most common changes to external IRB studies.
| Change | How to Initiate |
|---|---|
| Update UW PI | See Change Study PI [4] Instructions |
| Update Study Team (required to change access to the Zipline application) | See Update Study Team [5] Instructions |
| Change Reviewing IRB | Contact hsdrely@uw.edu [3] to initiate this process |
| Planning to Implement E-Consent | See Update Study Details instructions [6]
NOTE: Multi-site external IRB studies (includes most studies created after 1/7/2020) may also need to Create Site Modification [7] |
| Report Study Closure | Add a comment [8] to the study indicating that the study is now closed. Indicate that the IRB coordinator should receive a notification. |
Update Study Details
NOTE: Multi-site external IRB studies (includes most studies created after 1/7/2020) must submit a site modification to make changes to the local study team, update Local Funding Sources, or update the Local Site Documents.
1. In the study workspace, click Update Study Details
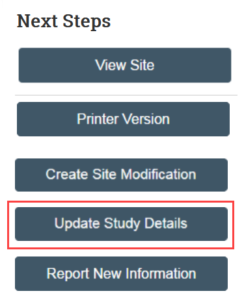
2. On the Study Update Information SmartForm, provide a summary of the update (for example, adding e-consent)
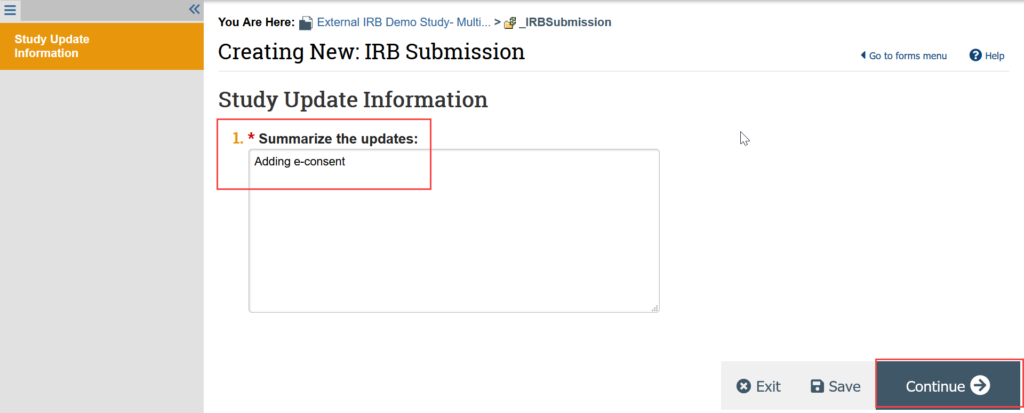
3. You are now in a draft version of the study SmartForm and should be on the Basic Study Information page. Update the draft version of the study to reflect any changes being made by making all needed revisions to the SmartForms and uploading any revised or new documents.
ADDING E-CONSENT:
- Update the revised REQUEST: External IRB Review [9] form on the External IRB SmartForm page. If you completed a REQUEST: External IRB Review that did not include questions about E-Consent, you will need to download and complete a new REQUEST: External IRB Review [9] form.
- If needed, upload the SUPPLEMENT: Other REDCap Installation [10] or TEMPLATE: Other E-Signatures Attestation Letter [11] on the Local Site Documents page. If the Local Site Documents page is not available, your study is a multi-site external study and you must submit a separate site modification [7] to add the SUPPLEMENT: Other REDCap Installation [10] and/or TEMPLATE: Other E-Signatures Attestation Letter [11].
-
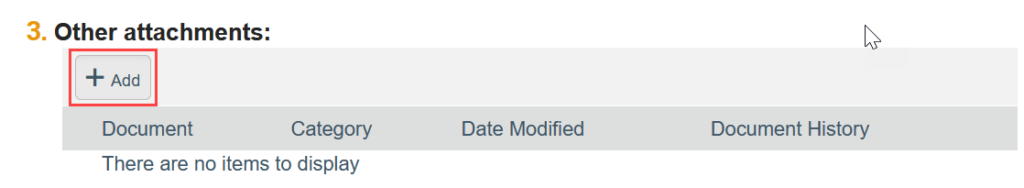
- To Add a Document:
-
-
-
- Click Add
- Select the new document from your computer
- Click OK in the Edit Attachment window
-
-
-
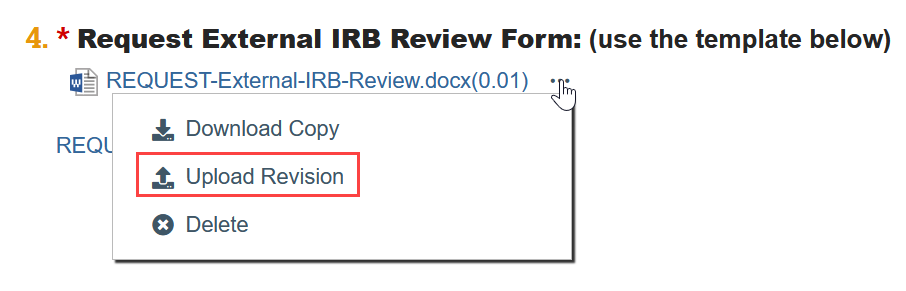
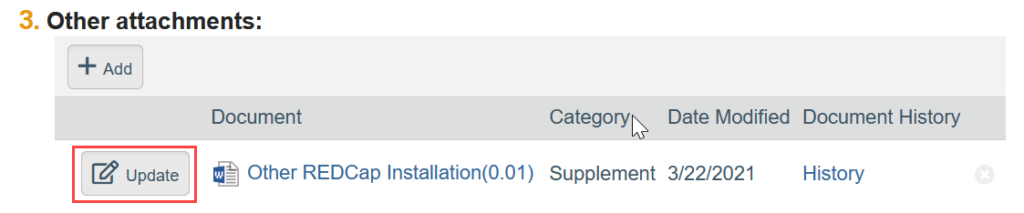
- To Update a Document:
-
-
- If needed, click the ellipsis by the document
- Click Upload Revision or Update
- Select the revised file from your computer
- Click OK in the Edit Attachment window
-
-
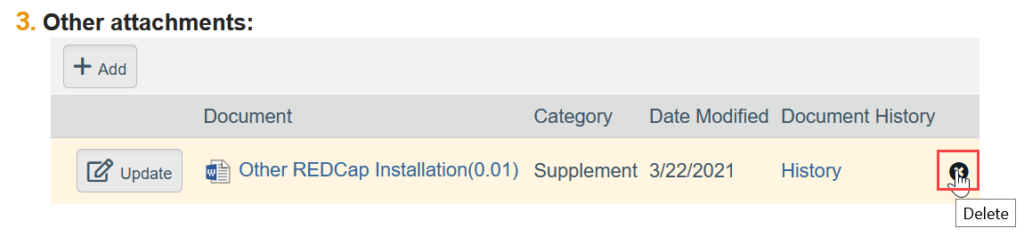
- To Remove a Document:
-
-
- Click the X by the document
-
4. Click Save and Exit out of the draft SmartForm once your changes are complete
5. Email hsdrely@uw.edu [3] when the update is complete. HSD staff will finalize the update after your change has been assessed.
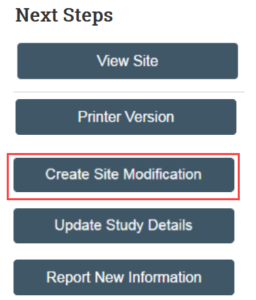
Create Site Modification
NOTE: This activity is only available for multi-site external studies (includes most external studies created after 1/7/2020).
1. In the study workspace, click Create Site Modification
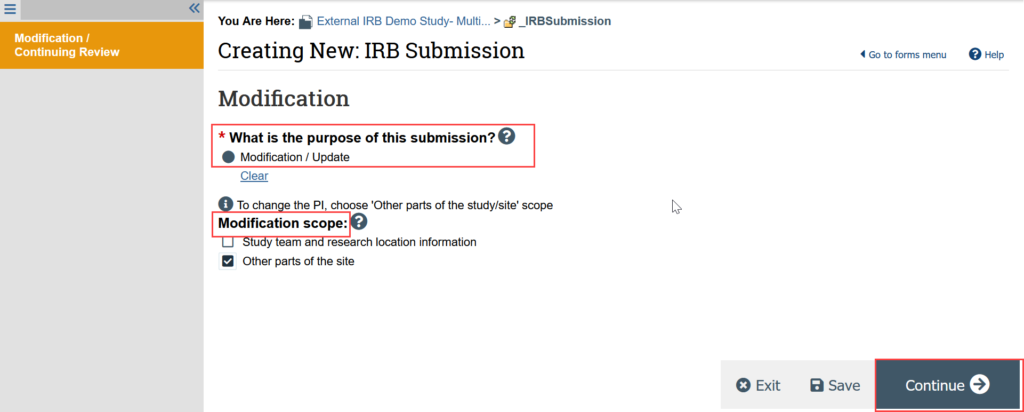
2. Indicate that the purpose of the submission is Modification/Update
3. Indicate the modification scope (can select both options)
-
- STUDY TEAM CHANGES: See Update Study Team [5] for detailed instructions
- ADDING E-CONSENT: Select “Other Parts of the Site”
4. Complete the Modification Information page and click Continue
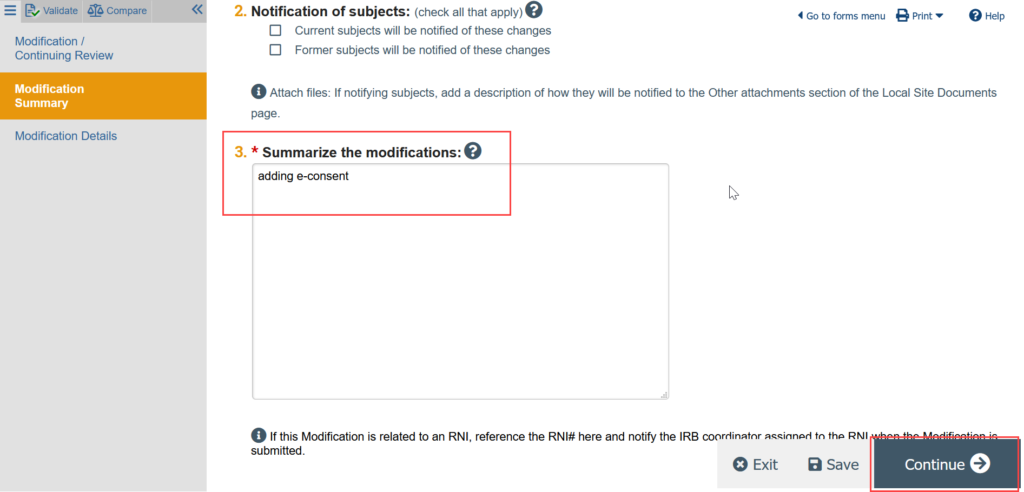
5. You are now in a draft version of the site SmartForm and should be on the Basic Local Site Information page. Update the draft version of the site to reflect any changes being made by making all needed revisions to the SmartForms and uploading any revised or new documents.
ADDING E-CONSENT:
- A site modification is only needed if you need to upload the SUPPLEMENT: Other REDCap Installation [10] and/or TEMPLATE: Other E-Signatures Attestation Letter [11] on the Local Site Documents page.
- If you haven’t already done so, Update Study Details [6] to update the revised REQUEST: External IRB Review [9] form on the External IRB SmartForm page.
To Add a Document:
- Click Add
- Select the new document from your computer
- Click OK in the Edit Attachment window

To Update a Document:
- If needed, click the ellipsis by the document
- Click Upload Revision or Update
- Select the revised file from your computer
- Click OK in the Edit Attachment window
To Remove a Document:
- Click the X by the document

6. Click Save and Exit/Finish to exit the SmartForm
7. Complete the Submit activity to send the modification to HSD for review
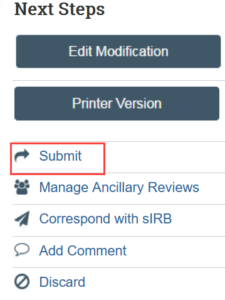
NOTE: The PI or a PI proxy [12] must complete this step.