Site submissions are only created for collaborative or multi-site studies where the UW is reviewing the activities of one or more other institution. The site submission only addresses the involvement of the relying institution, and it is approved separately from the overall study. The overall study must be approved before the relying sites can be activated. However, HSD staff may begin establishing the reliance agreements between the UW and each site while the study is being reviewed.
To learn more about when UW should review for other involved institutions, see Is the UW IRB the Right IRB? [2] Contact hsdrely@uw.edu [3] with any questions.
Watch this video for a short overview on adding a new site to an approved multi-site or collaborative study in Zipline:
Table of Contents
Initial Steps [4]
Completing the SITE Submission [5]
Submit SITE and Communicate with HSD [6]
Site Activation [7]
Initial Steps
Researcher Requests Addition of Sites
The process to request that HSD add a site that is relying on the UW for IRB review begins with the study application. Make sure that you’ve indicated that the UW should review for other institutions [8] on the study Basic Information SmartForm. Additionally, you’ll need to fill out the SUPPLEMENT Multi-site or Collaborative Research [9] and attach it to the study application in Zipline.
HSD Creates Sites in Zipline
In contrast to the overall study submission, which is created in Zipline by the study team, the site submission shell is created by HSD staff after the overall study is approved. Once the study, or modification requesting the addition of sites, is approved in Zipline, HSD will create a Zipline site submission shell for each site listed in the SUPPLEMENT Multi-site or Collaborative Research [9]. You will be notified when the sites submission shells have been created so that you can begin work on the SITE submissions.
Researcher Completes and Submits Sites for Review
HSD recommends that you prepare your site-specific materials before completing the Zipline SITE submission [5]:
- SUPPLEMENT Site Application (Contact) [10] or SUPPLEMENT Site Application (No Contact) [11], depending on the nature of the research; and
- Any additional site-specific materials, such as a site-specific consent form.
Completing the SITE Submission
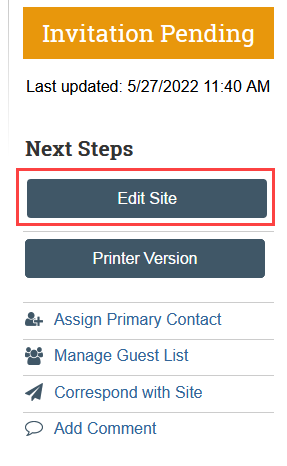
1. Navigate to the SITE submission in Zipline
- In the approved Zipline STUDY, go to the Sites tab and click the name of the site
- NOTE: SITE submissions will be in Invitation Pending state and so will not appear in My Inbox

2. Click Edit Site to complete the SmartForms and upload required documents
- Answer the questions on the Basic Information SmartForm in the SITE.
- Provide any site-specific funding information on the Local Funding Sources SmartForm. Note that this should not duplicate the overall study funding.
- Upload the site-specific documents to the Local Site Documents SmartForm, including the SUPPLEMENT Site Application and any site-specific materials, such as the site-specific consent form
Submit SITE And Communicate with HSD
Because the site submission workflow in Zipline does not include a formal clarification request loop like most other submission types, HSD will use comments to communicate back and forth with researchers about their SITE submissions. The site submission should remain in the Invitation Pending state throughout the review.

1. When the materials are ready for review, notify HSD by adding a comment to the SITE submission and notifying the IRB Coordinator
- In Next Steps, select Add Comment
- Enter your message to HSD in the text box
- Indicate that the IRB Coordinator should receive a notification
- Select OK

2. If you need to take additional action, HSD will notify you by adding a comment to the SITE submission
- You will receive an email notification that there is a comment on your SITE submission. Click the link in the notification to go directly to the submission and review the message from HSD.
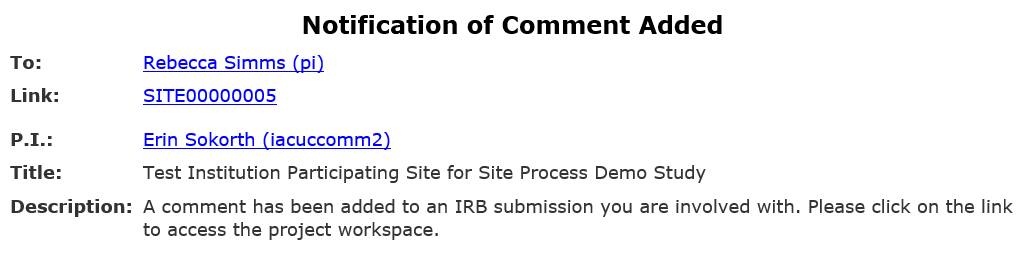

- Edit the SITE submission based on HSD’s comment
- When the materials are ready for review, notify HSD by adding a comment to the SITE submission

Site Activation
When a site is activated, the UW PI, PI proxies, and primary contact receive an email notification. The UW study team is responsible for notifying the site and delivering approved materials.
See Managing Multi-site Studies [12] for more information on continuing review, closure, and modifications.