December 10, 2009
Mechanism discovered by which body’s cells encourage tuberculosis infection
Scientists have discovered a signaling pathway that tuberculosis bacteria use to coerce disease-fighting cells to switch allegiance and work on their behalf. Epithelial cells line the airways and other surfaces to protect and defend the body. Tuberculosis bacteria co-opt these epithelial cells into helping create tubercles: the small, rounded masses characteristic of TB. The tubercles enable the bacteria to expand their numbers and spread to other locations.
By inciting parts of the immune system to go into overdrive, this same molecular signaling pathway may play other roles in inflammatory conditions such as arthritis and some forms of heart disease and cancer
“If we could keep this pathway from inciting the host immune system, we may be well on the way to finding innovative new therapies against TB, as well as other serious disorders,” said the senior researcher on the study, Dr. Lalita Ramakrishnan, University of Washington associate professor of microbiology, medicine and immunology. The results appear in the Dec. 10, 2009, express edition of Science.
Global health researchers are eager for new treatments for TB because many strains worldwide have become resistant to standard antimicrobials. Blocking a host pathway that the bacteria use would be an entirely different approach, Ramakrishnan explained, because it would keep the body from allowing the infection to take hold and be sustained, rather than a treatment aimed at killing the bacteria themselves. A host pathway blocker, if one becomes available, might also be quicker than current therapies, which take a long time to subdue the TB infection.
“Most diseases, such as high blood pressure and depression, are already being treated by blockers and inhibitors of host enzymes and pathways,” Ramakrishnan noted, “Many of these turn down certain cell signals as part of their therapeutic action. We and some other researchers are now exploring the possibility of blocking or inhibiting molecular mechanisms in the body to prevent or treat infectious diseases as well. ”
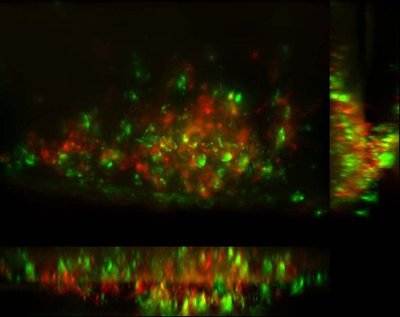
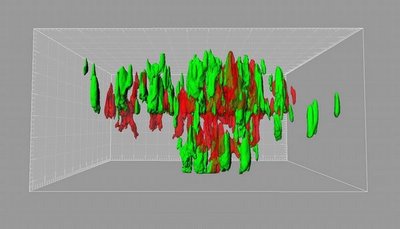
Earlier studies in the zebrafish by the Ramakrishnan lab demonstrated that TB tubercles were not, as previously thought, the way that the body walls off the bacteria to protect itself. Instead, these nodules (also called granulomas) are hubs for bacteria production and distribution. Uninfected macrophages — the body’s frontline soldiers that can eat and destroy many bacteria — are recruited to the nodules, where they become TB-infected. However, the TB bacteria are able to grow in the macrophages, rather than being killed, likely by dampening the macrophages’ defenses.
So by wooing more macrophages into the granuloma, the bacteria can use them to expand further. Some germ-laded macrophages then move to a new location, where they again attract more macrophages. New tubercles form and the scene is repeated.
Ramakrishnan and her research team have identified a molecular mechanism by which the mycobacteria that cause TB induce the body to form these production and distribution nodules. Researchers have long known that TB virulence is associated with a small protein the bacteria secrete, called ESAT-6.
Ramakrishan’s group now has found that this secreted bacterial protein induces epithelial cells — the cells that make up membranous tissue covers inside the body — to produce an enzyme called MMP9. This enzyme has many functions including breaking down gelatin — a connective tissue protein — into its components. In people, the presence of MMP9 is associated with increased susceptibility to infection and worse outcomes. The findings of this new study explain why this might be the case. MMP9 is also implicated in the development of several non-infectious inflammatory conditions, like arthritis, as well as heart disease and cancer.
Epithelial cells were once thought to be bystanders as tuberculosis took hold, according to the research group. However, their latest findings suggest that secretion of MMP9 by epithelial cells is amplified in the vicinity of a single TB infected macrophage. The activity of this enzyme draws in uninfected macrophages to join the infected macrophage to form and expand the granuloma.
“TB bacteria may have a two-prong strategy,” said the first author of the Dec. 10 Science Express report, Dr. Hannah E. Volkman, who recently received her Ph.D. from the UW Molecular and Cellular Biology Program, “whereby the bacteria simultaneously suppress the macrophages inflammatory programs in order to create a hospitable niche inside them, while prodding epithelial cells to signal more macrophages to arrive and be unwitting participants in their home expansion project.”
The researchers genetically “knocked out” MMP9 production in zebrafish embryos to see if that made them more resistant to TB. After TB infection, these embryos indeed had greater survival rates, fewer bacteria, and fewer granulomas than their normal, MMP9-producing siblings. This finding suggested that intercepting the production of MMP9 in epithelial cells should be further studied as a possible TB therapy.
“These novel findings,” said Dr. William Parks, a UW professor of medicine and director of the UW Center for Lung Biology who was not part of this study, “point to new ways in which the body’s resident cells can effect an inflammatory response and may have relevance beyond TB infection. The pathogen-to-epithelium-to-macrophage pathway they uncovered should provide several new avenues that could be targeted for intervention.”
Co-authors of the article, “Tuberculous Granuloma Induction via Interaction of a Bacterial Secreted Protein with Host Epithelium,” in addition to Volkman and Ramakrishnan, are Tamara C. Pozos, a former UW infectious disease fellow who is now on the faculty of Children’s Hospital and Clinics of Minnesota; John Zheng, a UW medical student; J. Muse Davis, an M.D./Ph.D. student at Emory University; and John F. Rawls, assistant professor of cell and molecular physiology, microbiology, and immunology, University of North Carolina, Chapel Hill.
The study was funded by the Burroughs Wellcome Fund, Pew Scholars Program, National Institutes of Health, American Heart Association, Pediatrics Infectious Disease Society, Children’s Health Research Center, and a National Defense Science and Engineering fellowship
# # #