November 5, 2008
UW researchers find that having a big heart is not always a good thing
A cell signaling pathway that abnormally promotes heart muscle cell growth invariably leads to unhealthy enlargement of the heart. Researchers have recently discovered that the heart’s overproduction of a molecule known as A-kinase anchoring protein-Lbc amplifies these harmful signals.
These results, from a study led by Dr. John D. Scott, University of Washington (UW) professor of pharmacology, the Edwin G. Krebs – Speights Professor of Cell Signaling and Cancer Biology, and an investigator in the Howard Hughes Medical Institute, were published in the Oct. 24 edition of Molecular Cell.
Anchoring proteins keep heart enzymes in a particular spot, just as a ship is anchored to keep it from drifting. In doing so, the anchoring proteins speed up biochemical reactions that generate signals inside the cell. Anchoring proteins also bring together the molecules that start and stop the signal. They are complex, highly organized message coordinators.
The UW authors noted that biomechanical stress on the heart from high blood pressure or diseased valves can induce a cellular response that enlarges heart muscle cells. The UW cell-signaling researchers also pointed to studies by other scientists suggesting that catecholamines — hormones like adrenaline that give the body power and strength to face emergencies by making the heart beat faster and contract more easily — can harm the heart if they remain elevated for long periods of time.
Persistently high levels of catecholamines can evoke a signaling pathway that causes bigger heart cells. They do this by activating the myocyte [muscle cell] enhancing factor. This factor induces a re-programming of genes that produces a “fetal cell response.” Genes that were turned off after the heart had formed are turned on again. Heart muscle cell size then increases, along with other cellular changes.
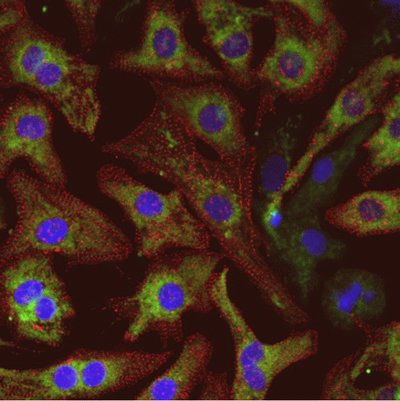
Scott and other UW researchers discovered an abundance of A-kinase anchoring protein-Lbc in these overgrown heart muscle cells. This finding supports results from other scientists who have suggested that the cellular processes controlled by the enzymes called protein kinases are switched on in patients with heart failure. Scott and his colleagues were able to show that active kinases become more concentrated in the cell nucleus when the anchoring protein is around. Their presence in the nucleus becomes a problem because they inappropriately turn on genes that drive heart muscle cells toward a disease state.
Using advanced microscopy methods like live-cell imaging, fluorescent kinase activity reports and RNA interference techniques, Scott and his colleagues were able to observe the movement of proteins inside heart cells with increases in the amount of A-kinase anchoring protein-Lbc.
“We believe this is one of the things that go wrong in the early stages of congestive heart failure,” Scott said. “We are encouraged by this discovery and are poised to examine the pathology of this process in more detail. Because the A-kinase anchoring protein is more readily detected in diseased cells, it may eventually become a useful prognostic marker for early-stage heart disease.”
Scott noted that additional laboratory studies are needed to determine if A-kinase anchoring protein-Lbc is a biomarker that predicts the development of an enlarged heart, and to find out which genes respond to stimulus to increase production of this anchoring protein.
In addition to Scott, the researchers on the study were F. Donelson Smith, UW; Graeme K. Carnegie, Joseph Soughayer, Benjamin S. Pedroja, Fang Zhang, and Lorene K. Landeberg, all of the Howard Hughes Medical Institute and Vollum Institute at Oregon Health and Science University; Darlo Diviani of the University of Lausanne, Switzerland; Michael R. Bristow, of the University of Colorado Health Sciences Center, Denver; and Maya Kunkel and Alexandra Newton, both of the University of California, San Diego.
Grants from the Fondation Leducq and the National Institutes of Health supported the research.