Illustration by Kate Sweeney.


Illustration by Kate Sweeney.
Sanders' first task in the biomaterials project is to study the mechanics of healing. When a wound occurs, the body automatically launches an inflammatory response to summon extra cells to the wound site, which is why it becomes red and puffy. Then, directed by highly specific protein signals, these cells begin dividing to regenerate tissue and close the wound.
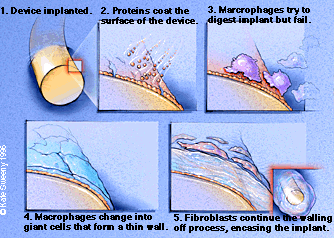
The same inflammatory response occurs when a medical device is implanted in the body. But the cells summoned to the wound site don't recognize the implant material. Instead of attaching to the device, the cells wall it off with scar tissue. This same reaction harmlessly occurs with bullets and pieces of shrapnel that can't be removed from the bodies of some war veterans. But with many medical implants, like artificial heart valves and cochlear implants, the scar tissue disrupts the device's performance and often requires another operation.
Sanders' work has given her a clear understanding of a critical healing problem associated with medical implants that breach the skin, such as catheters. Since the skin doesn't seal around the implant, the body's main barrier against bacteria is broken and these implants almost always become infected. The implant must be removed and can't be replaced until the infection is cleared up. Sometimes the patient isn't able to wait that long.
In addition to studying the healing process, Sanders' team will devise ways to test new biomaterials developed by UWEB. In collaboration with dermatology researchers, Sanders' research group--which includes seven students, most of whom are undergraduates--already has figured out how to keep a small swatch of pig skin alive in the laboratory. This will allow researchers to test new biomaterials in living tissue.
"It's a very controlled situation, but it's a good first step," Sanders says.
Tom Horbett, professor of bioengineering and chemical engineering, is an expert on how proteins and cells interact with foreign materials in the body. He will head up the project's cell biology team. He has been working literally side-by-side with Ratner on medical implants for a quarter of a century. Horbett's research group has two missions: to understand how cells and proteins communicate to prompt the body's hostile reaction to foreign materials and then to figure out how to control those signals to induce normal healing.
Scientists know that proteins direct and carry out most cellular functions in the body, including healing. When doctors put a medical device in the body, for example, it is immediately covered with a layer of proteins that identify the implant as a foreigner. The proteins begin communicating with surrounding cells. These signals, Horbett explains, are believed to stimulate certain "problem cells," which then form the tissue that encapsulates the implant.
Horbett's team hopes to identify the proteins that "turn on" the encapsulation process so they can try to keep those proteins turned off. The group's second major focus is to identify another set of key protein fragments, or peptides, that cells attach to in normal tissue. The plan is to coat implants with these peptides. Horbett wants to fool cells into thinking the implant is normal tissue so that they attach to the devices rather than build scar tissue around them.
"We'll be pioneers when we get to that point," Horbett says. "Other people have thought about using peptides to modify cell responses, but nobody has tried using peptides to prevent encapsulation."
One Molecule at a Time
Denice Denton: A New Kind of Dean
Send a letter to the editor at columns@u.washington.edu.